
Is NO2+ (Nitronium Ion) Polar or Nonpolar? YouTube
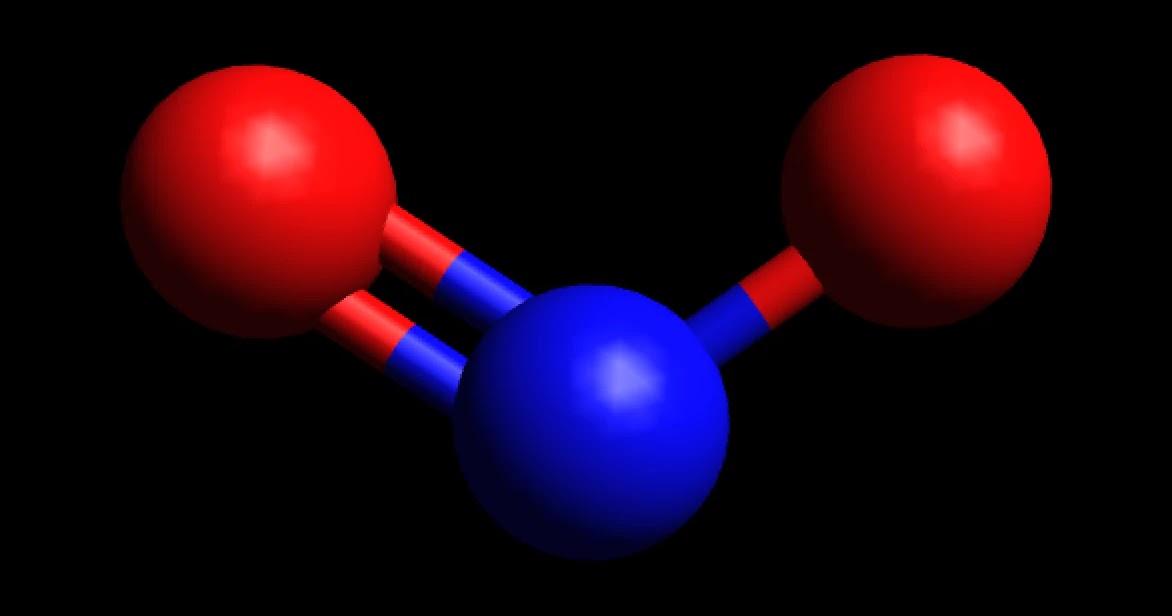
Nitrogen dioxide is a chemical compound with the formula NO 2.One of several nitrogen oxides, nitrogen dioxide is a reddish-brown gas.It is a paramagnetic, bent molecule with C 2v point group symmetry.Industrially, NO 2 is an intermediate in the synthesis of nitric acid, millions of tons of which are produced each year, primarily for the production of fertilizers.
SOLVED Which molecule has polar bonds but is nonpolar? A. ClF3 B. H20
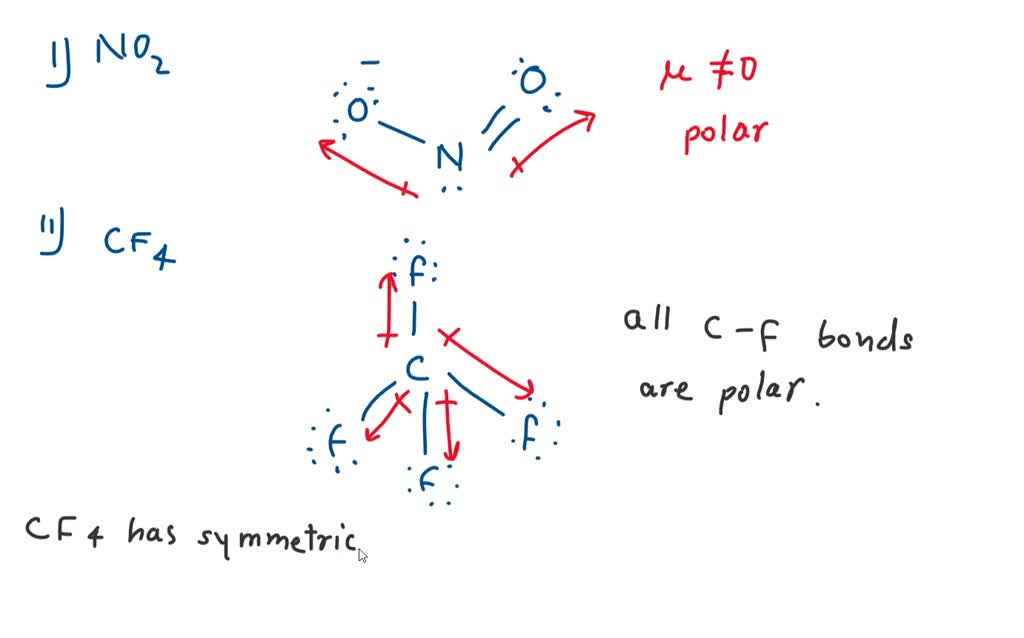
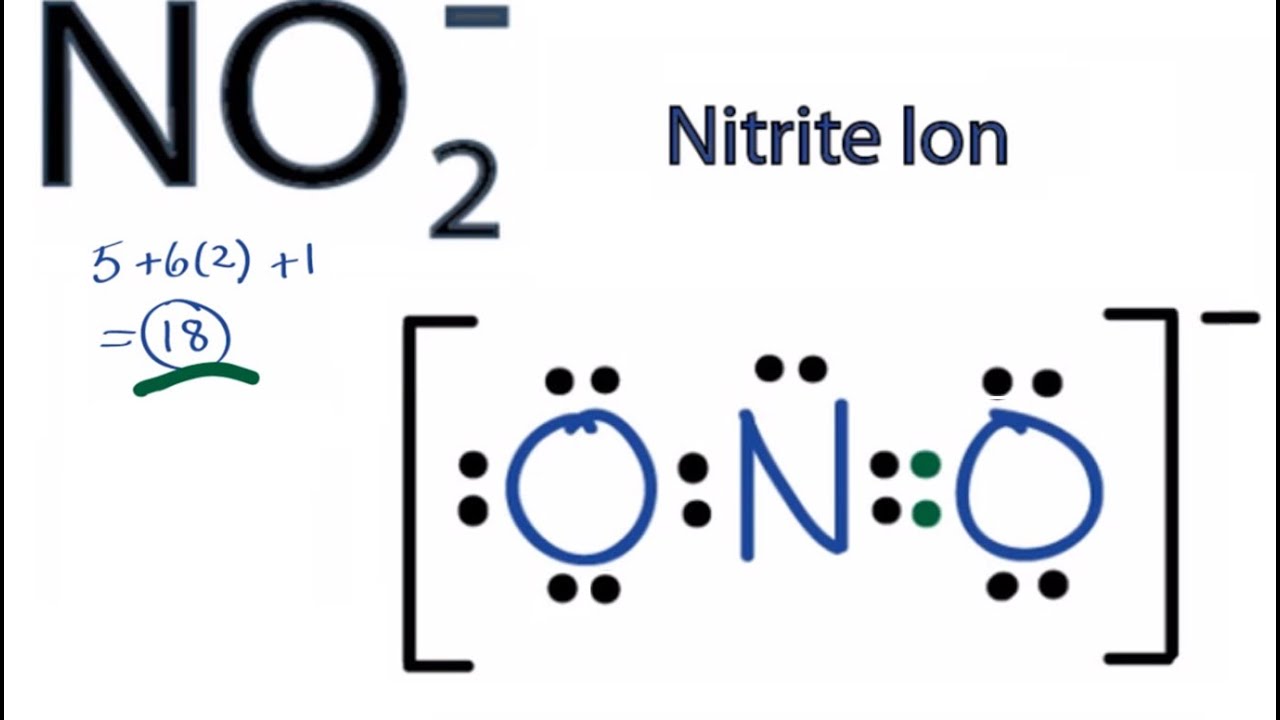
Nitrite [NO2]- is a polar molecular ion. It consists of a nitrogen (N) atom and two oxygen (O) atoms. The nitrogen atom is present at the center of the molecular ion, having a lone pair of electrons. In contrast, two oxygen (O) atoms occupy terminal positions, one on each side of the central N-atom, thus forming a bent shape.

Is NO2+ Polar or Nonpolar? YouTube
Therefore, a N-O bond is polar with the oxygen negative and the nitrogen positive. If the NO2 molecule were linear, the polar bonds would oppose each other and the molecule would be non-polar. Instead, a non-bonding pair of electrons are on nitrogen. The pair of electrons repel the bonds and distort the molecule into a bent geometry.

Is N2O Polar or Nonpolar? YouTube
Table 13.5. 1: Bond Polarity. If the difference between the electronegativities of the two atoms is small, neither atom will take the shared electrons completely away from the other atom, and the bond will be covalent. This is the case when two nonmetals are bonded to each other.

Properties of NO2+
Learn to determine if NO2 + is polar or nonpolar based on the polarity between bonds and the molecular geometry (shape).Ions, like NO2+ are sometimes confusi.
No2 Lewis Structure
NO2 is a polar molecule and the Oxygen atom closest to negative side as the electronegativity of Oxygen (3.44) is comparatively greater than Nitrogen (3.04) so that Nitrogen has a partial positive charge and Oxygen has a partial negative charge established within the molecule.

NO2 é polar ou apolar
NO2+ is a nonpolar molecule despite two N-O bonds are polar. NO2+ has a linear geometry due to no lone pair of electrons on central N atom which causes cancellation of both positive and negative charges produced on the molecule, as a result, the net dipole becomes zero.

Table of Contents
1. Another non polar molecule shown below is boron trifluoride, BF 3. BF 3 is a trigonal planar molecule and all three peripheral atoms are the same. Figure 4.12.1 4.12. 1 Some examples of nonpolar molecules based on molecular geometry (BF 3 and CCl 4 ). Polar molecules are asymmetric, either containing lone pairs of electrons on a central atom.

Is Nitrogen Dioxide (NO2) Polar or NonPolar? Lewis Structure YouTube
Figure 4.4.1 4.4. 1 Polar versus Nonpolar Covalent Bonds. (a) The electrons in the covalent bond are equally shared by both hydrogen atoms. This is a nonpolar covalent bond. (b) The fluorine atom attracts the electrons in the bond more than the hydrogen atom does, leading to an imbalance in the electron distribution. This is a polar covalent bond.

Polar Bear No2
Welcome to the Mentor Center. In this video, I find out if N2O (nitrous oxide/laughing gas) is polar or not.**MISTAKE** I realized that the electrons were su.

Is NO2+ Polar or Nonpolar?
NO2 Polar or Nonpolar To determine if NO 2 is polar or nonpolar, we need to first determine its geometry. This presumes knowing the rules for drawing a correct Lewis structure and you can find more details about Lewis structures here. Nitrogen is the central atom, so we can draw the skeletal structure:

Is NO2 Polar or Nonpolar? [Brief Explanation in simple terms]
Who has not heard about NO2, Nitrogen Dioxide? It is one of the most common gaseous molecules having a reddish-brown hue. It can be cooled and compressed into a yellowish-brown liquid for tendshipping and transport. A highly toxic poisonous chemical compound, NO2 is a major air pollutant and belongs to the group of oxides of nitrogen.
MakeTheBrainHappy Is NO2 Polar or Nonpolar?
So, Is NO2+ Polar or Nonpolar? NO2+ (Nitronium ion) is nonpolar in nature because it has a linear geometrical structure due to which polarity of opposite NO bonds gets canceled by each other resulting in the nonpolar NO2+ ion. Nitronium ion is a stable ion in normal conditions.

NO2 Molecular Geometry / Shape and Bond Angles YouTube
Carvone oxide, cis-Piperitenone oxide; Other names: trans-Carvone oxide; Carvone-1,2-epoxide; carvon-1,2-oxide; trans-Carvone epoxide; 1-methyl-4-(1-methylvinyl)-7-oxabicyclo[4.1.0]heptan-2-one Permanent link for this species. Use this link for bookmarking this species for future reference. Information on this page: Gas Chromatography.

Is NO2+ Polar or Nonpolar Science Education and Tutorials
Bond Polarity Calculator Calculate the molecular polarity (polar, non-polar) of a chemical bond based on the electronegativity of the elements. First Element Second Element Calculate Bond Polarity How To Calculate Bond Type and Polarity The bond polarity between two atoms can be estimated if you know the electronegativity of both elements.
[DIAGRAM] Dot Diagram Of N2o
Learn to determine if NO (Nitric oxide) is polar or non-polar based on the Lewis Structure and the molecular geometry (shape).We start with the Lewis Structu.