Modelo Atomico Del Magnesio Compuesto
Magnesium is an essential part of the human body and it is required for biochemical reactions in the body. Magnesium is the 11th abundant element (by mass) found in the human body. Out of the total amount of magnesium present in the human body, around 60% of magnesium is in the skeleton, 39% is in muscle tissues and 1% being extracellular.

Magnesium Atomic Number Atomic Mass Density of Magnesium
While protons and neutrons are located inside the nucleus at the center of the atom, electrons are located outside the nucleus in what is often called the electron cloud. Figure 4.4.1 4.4. 1: Electrons are much smaller than protons or neutrons. If an electron was the mass of a penny, a proton or a neutron would have the mass of a large bowling.

Symbol and electron diagram for Magnesium Vector Image
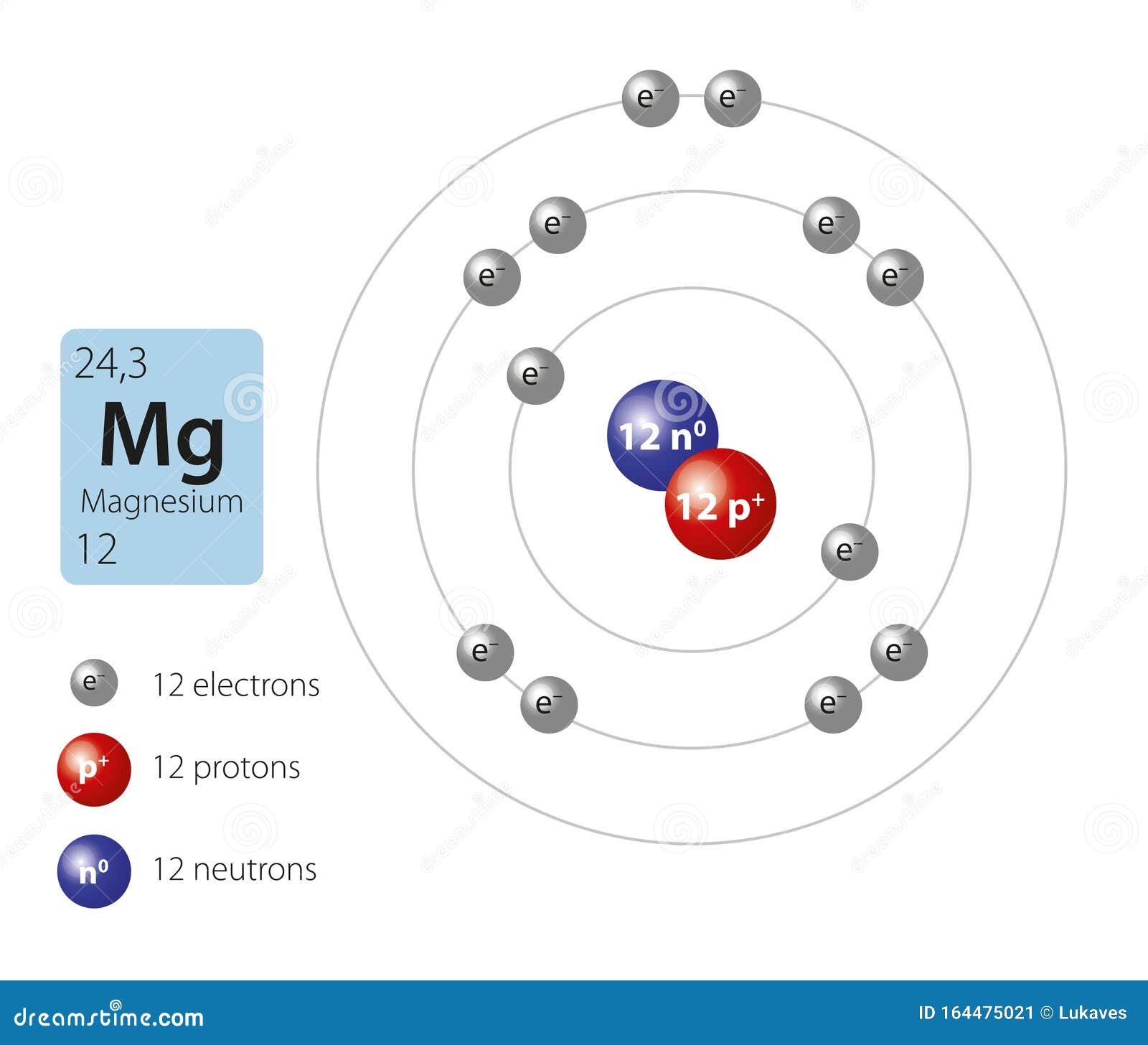
Summary Atomic Number - Protons, Electrons and Neutrons in Magnesium Magnesium is a chemical element with atomic number 12 which means there are 12 protons in its nucleus. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z.

How To Find The Number Of Neutrons In An Element redesigngreece
An isotope is a compound with the same number of protons and electrons, but different number of neutrons. The three most natural isotopes of Mg are Mg-24, Mg-25, and Mg-26. Mg-24 (12 neurtrons) is 78.9%, Mg-25 (13 neutrons) is 10% and Mg-26 (14 neutrons) is 11.01%, of all the Magnesium found in nature.
Magnesium element with symbol Mg and atomic number 12.isolated
Magnesium is a chemical element with atomic number 12 which means there are 12 protons in its nucleus. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs.
Magnesium Protons Neutrons Electrons Electron Configuration
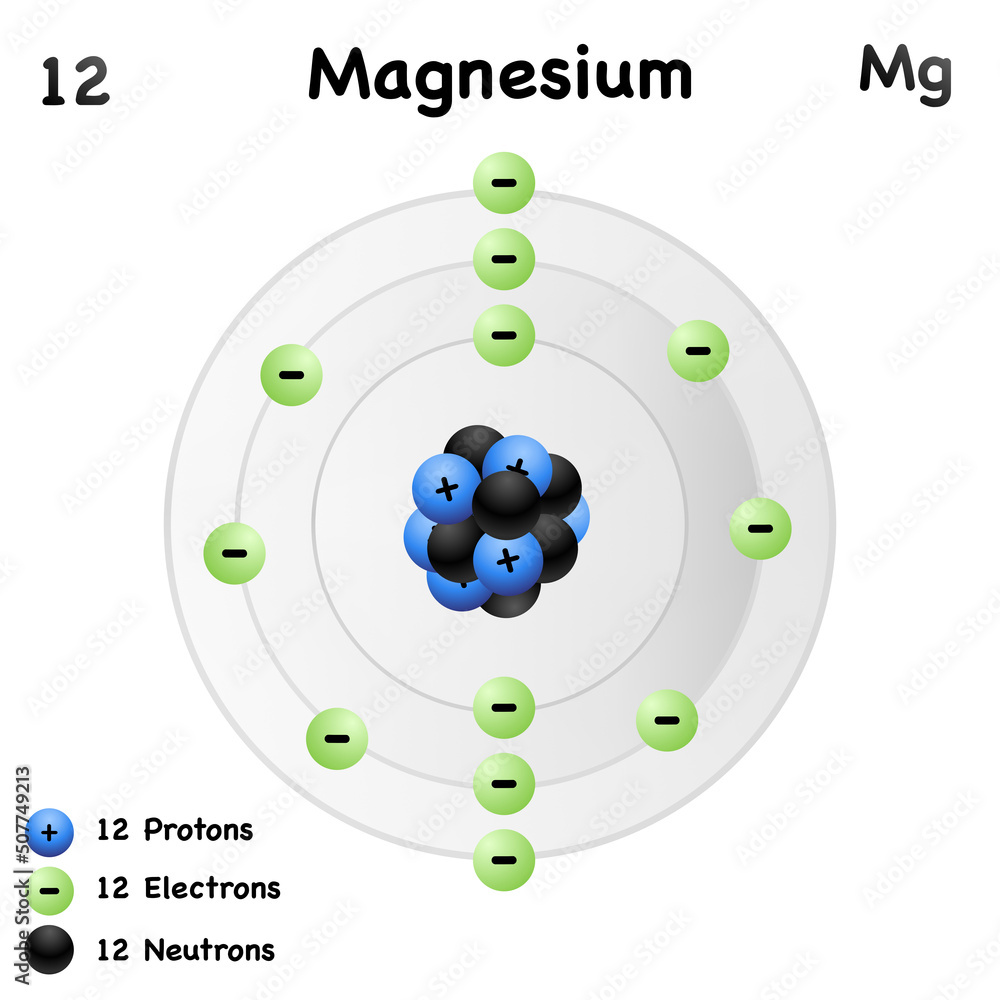
Neutrons = atomic mass - atomic number. Magnesium neutrons. The atomic mass of magnesium is 24.305, so we'll take the roundup value as 24. And the atomic number of magnesium is 12. Subtract the atomic number (12) from the atomic mass (24). Hence, magnesium has a total of 24 - 12 = 12 neutrons.
subatomic particles Montessori Muddle
Table 2.2 The number of protons in the nucleus of an atom is its atomic number (Z). This is the defining trait of an element: Its value determines the identity of the atom. For example, any atom that contains six protons is the element carbon and has the atomic number 6, regardless of how many neutrons or electrons it may have.
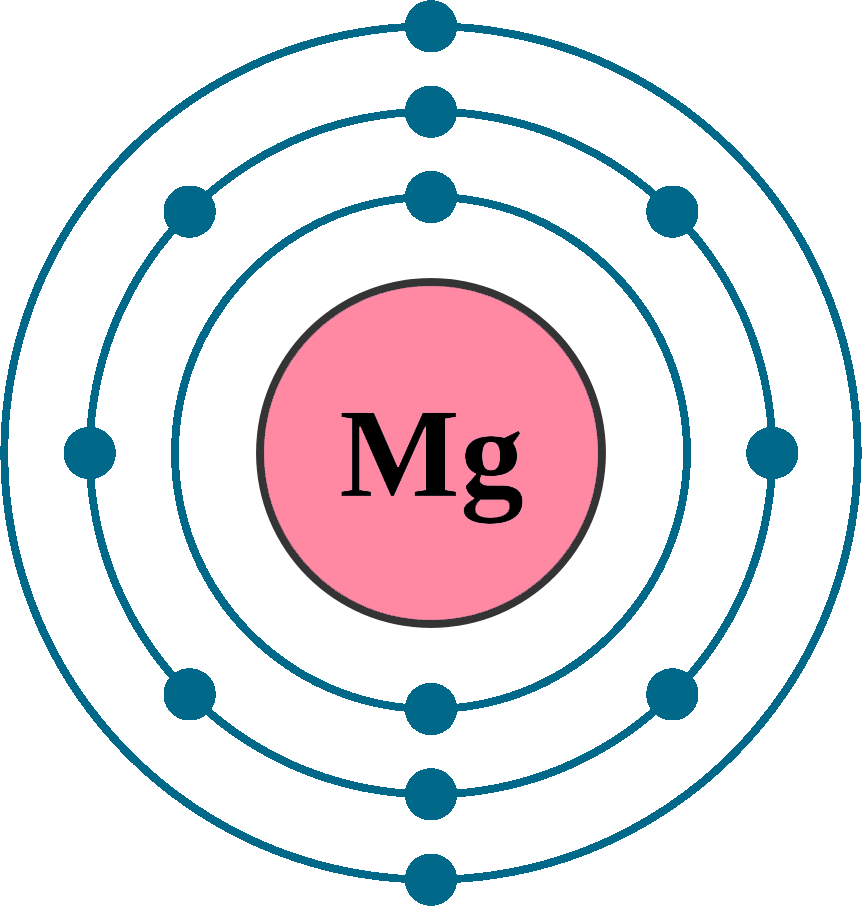
magnesium electron configuration Newton Desk
That is, magnesium is a cation element. Mg - 2e - → Mg 2+. The electron configuration of magnesium ion (Mg +2) is 1s 2 2s 2 2p 6. This electron configuration shows that magnesium ion (Mg +2) has acquired the electron configuration of neon and it achieves an octave full stable electron configuration.
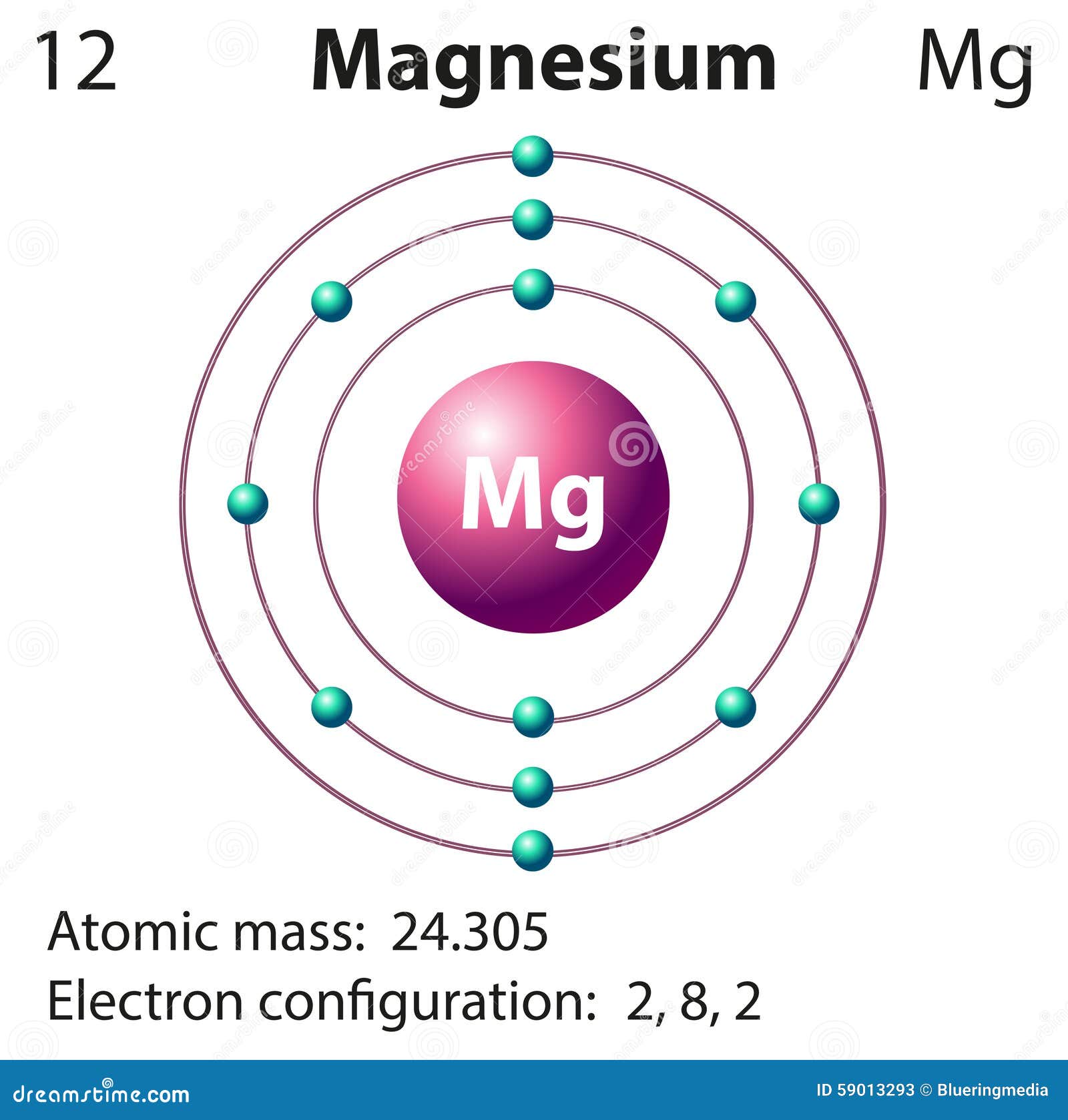
Diagram Representation Of The Element Magnesium Stock Vector Image
Learn how to calculate the molar mass of Mg(OH)2, a common compound used in antacids and laxatives, by using the periodic table and a simple formula. This video tutorial will show you the steps.
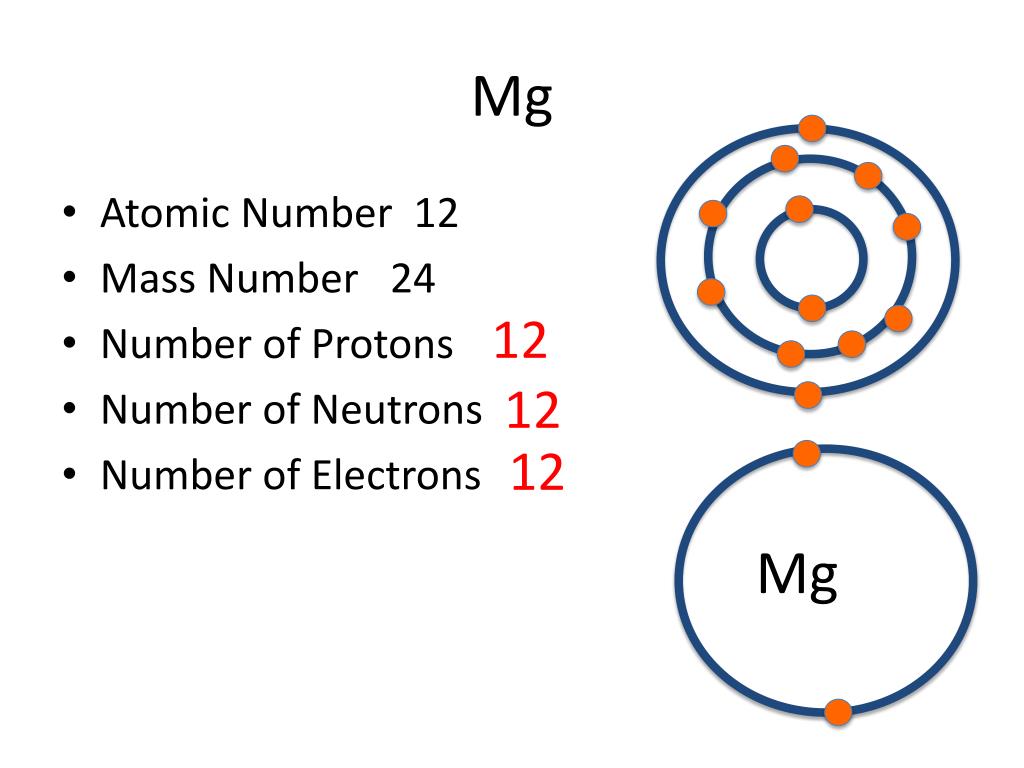
Mg Periodic Table Protons Neutrons And Electrons Awesome Home
27 protons, 33 neutrons, 27 electrons. The cobalt atom has 27 protons as seen in the periodic table. There are also 27 electrons because the charge is 0. There are 33 neutrons because 60-27 = 33. 3 H. 1 protons, 2 neutrons, 1 electrons. There is 1 proton because the element is hydrogen. There is 1 electron because the atom is neutral. There are.
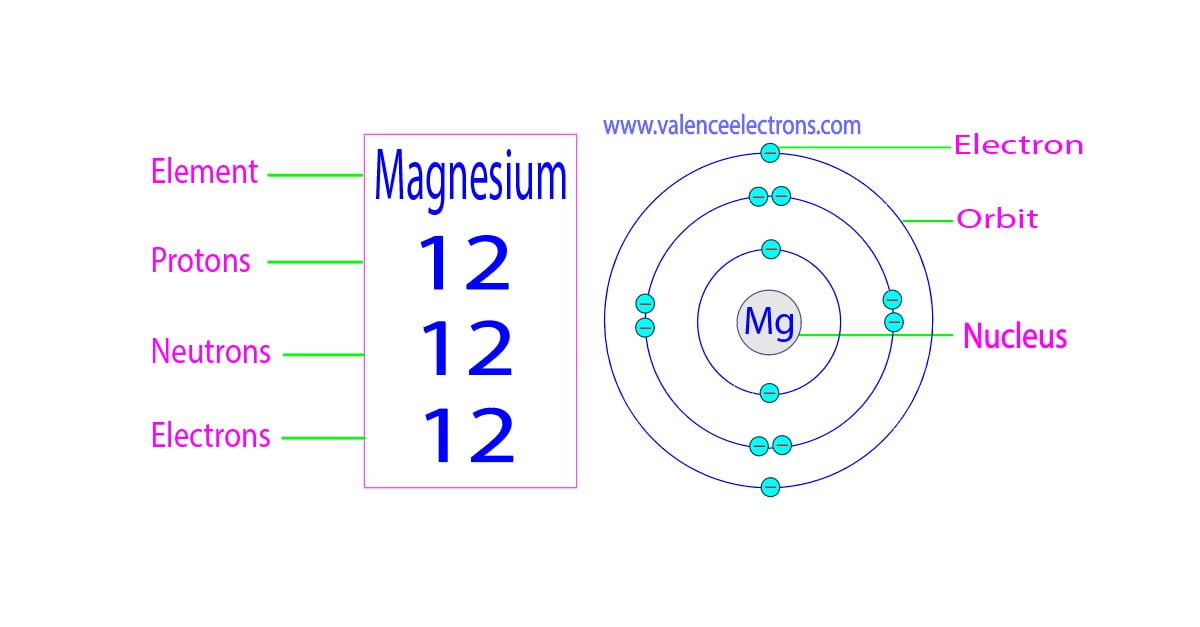
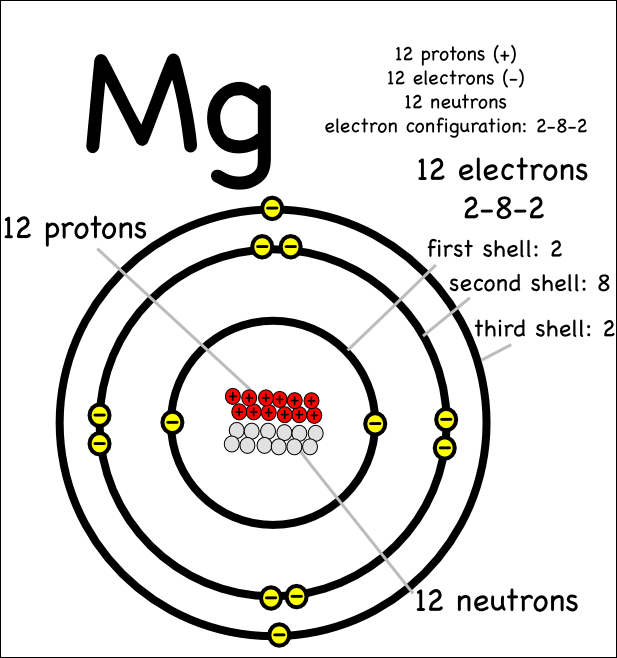
Protons, Neutrons, Electrons for Magnesium (Mg, Mg2+)
Magnesium is the 12th element of the periodic table so its atomic number is 12. The atomic number of an element is equal to the number of protons and electrons in that element. Therefore, a magnesium atom has twelve protons and twelve electrons.

Magnesium Electron Configuration (Mg) with Orbital Diagram
There are 12 protons and 10 electrons in a "Mg"^(2+) ion, the normal amount of neutrons is 12. Magnesium is an element with atomic number 12. This means that every magnesium atom will have 12 protons. In a magnesium atom, there are 12 electrons, to make the atom have a neutral charge. When an ion is formed, the magnesium atom loses electrons. The 2+ charge shows that the magnesium ion has two.

Magnesium, atomic structure Stock Image C018/3693 Science Photo Library
The most common and stable type of magnesium atom found in nature has 12 protons, 12 neutrons, and 12 electrons (which have a negative charge). An image of the secondary beam "cocktail" produced at a cyclotron center in Japan for a study of Mg-40, an exotic isotope of magnesium.
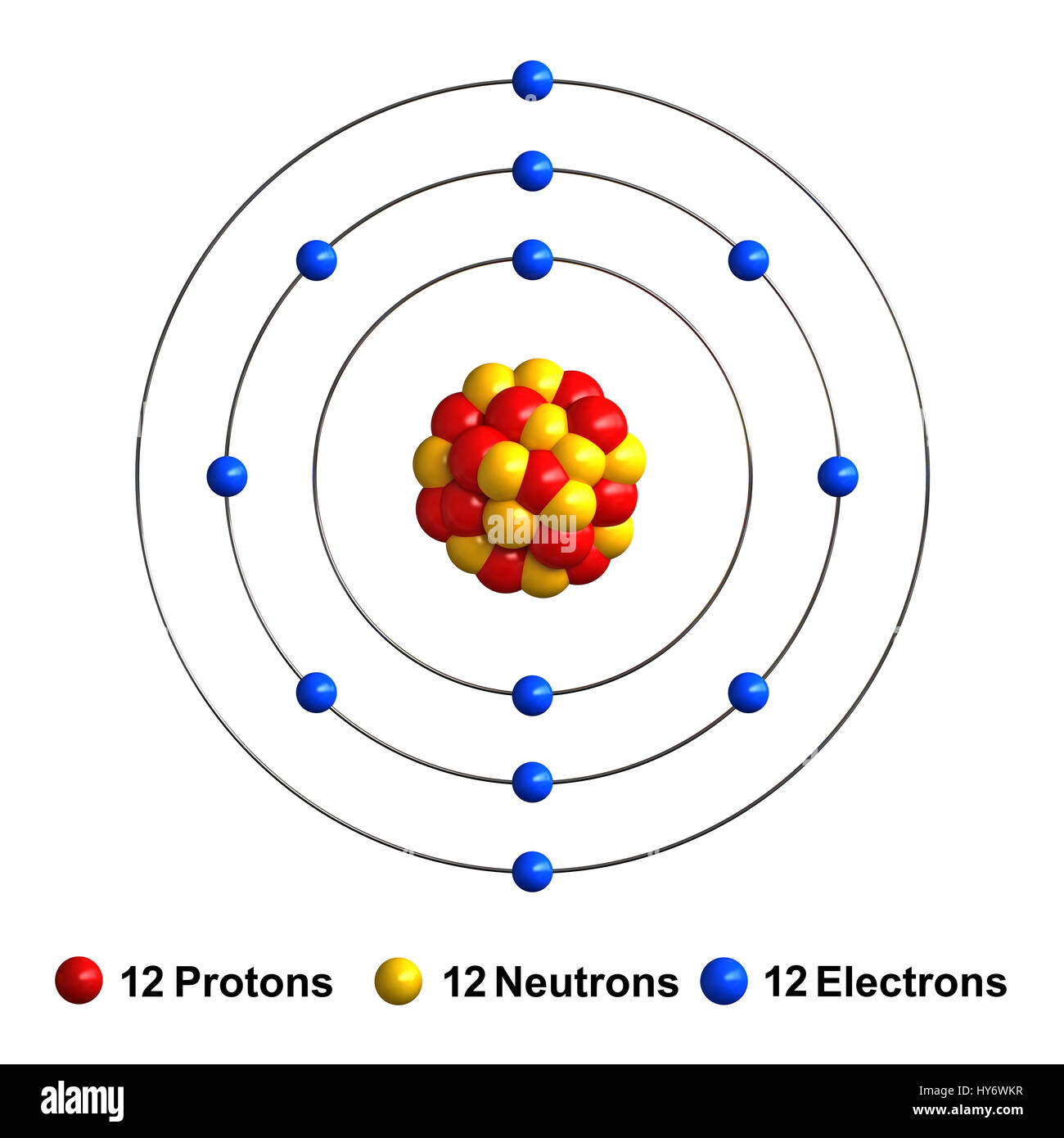
3d render of atom structure of magnesium isolated over white background
Magnesium has 12 protons, 12 neutrons and 12 electrons. But how will you find the number of protons, neutrons and electrons in Magnesium (Mg)? Well, it is very easy to find the protons, neutrons and electrons of magnesium atom. Here I have given a very simple method for finding the protons, neutrons and electrons of magnesium atom.

Magnesium Periodic Table and Atomic Properties
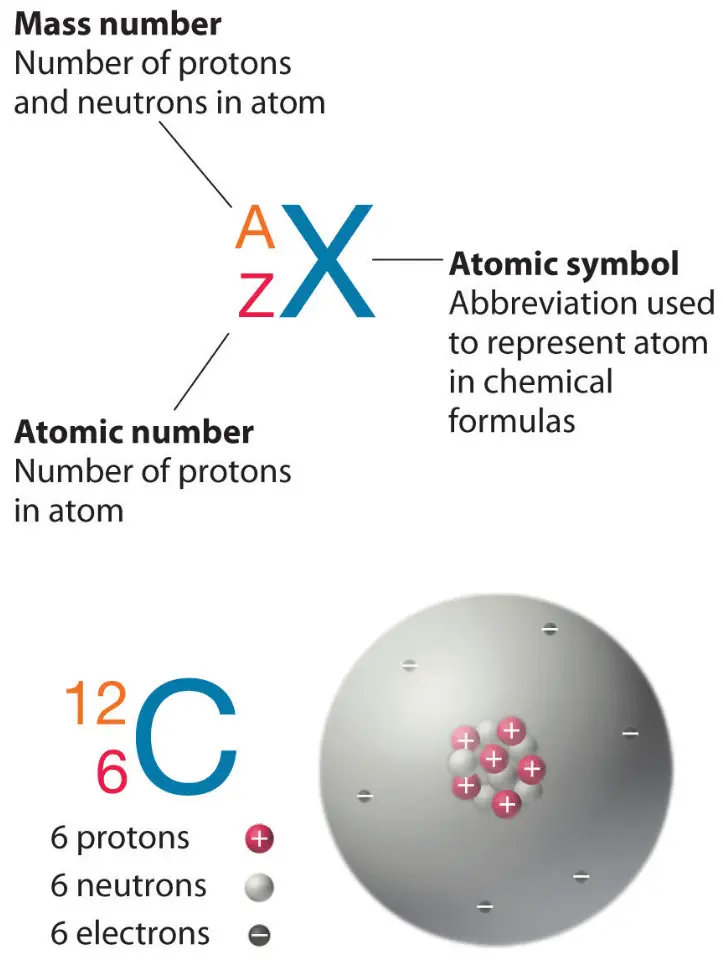
Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74). Since the iodine is added as a 1− anion, the number of electrons is 54 [53 - (1-) = 54]. Exercise. An ion of platinum has a mass number of 195 and contains 74 electrons.
Magnesium Periodic Table and Atomic Properties
Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74). Since the iodine is added as a 1− anion, the number of electrons is 54 [53 - (1-) = 54]. Exercise 2.3.1. An ion of platinum has a mass number of 195 and contains 74 electrons.