[Solved] draw a lewis structure for the followings HBr C2HCI Course Hero
Write Lewis structures for the following:(a) H2(b) HBr(c) PCl3(d) SF2(e) H2CCH2(f) HNNH(g) H2CNH(h) NO-(i) N2(j) CO(k) CN-OpenStax™ is a registered trademark.

Hbr Lewis Structure Transborder Media
Exercise 10.4.1 10.4. 1. Use Lewis electron dot diagrams to illustrate the covalent bond formation in Cl 2. Answer. When working with covalent structures, it sometimes looks like you have leftover electrons. You apply the rules you learned so far, and there are still some electrons that remain unattached.

Best Overview Is HBr Polar or Nonpolar? Science Education and Tutorials
Lewis structure: diagram showing lone pairs and bonding pairs of electrons in a molecule or an ion. Lewis symbol: symbol for an element or monatomic ion that uses a dot to represent each valence electron in the element or ion. lone pair: two (a pair of) valence electrons that are not used to form a covalent bond.
[Solved] draw a lewis structure for the followings HBr C2HCI Course Hero
Steps of drawing HBr lewis structure Step 1: Find the total valence electrons in HBr molecule. In order to find the total valence electrons in HBr (hydrogen bromide) molecule, first of all you should know the valence electrons present in a single hydrogen atom as well as bromine atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)

Lewis Dot Structure For Hbr
What is the lewis dot structure for hydrogen bromide (HBr)? The total number of electrons would 8. There is a single bond connecting hydrogen and bromine. Since hydrogen is satisfied by only two electrons, the rest of the lone pairs will end up on bromine. What is the lewis dot structure of carbon tetrachloride (CCl 4)? There are 32 valence.

How to draw HBr Lewis Structure? Science Education and Tutorials
The first step is to sketch the Lewis structure of the HBr molecule, to add valence electrons around the bromine atom; the second step is to add valence electrons to the one hydrogen atom, and the final step is to combine the step1 and step2 to get the HBr Lewis Structure.

Hbr Lewis Structure Transborder Media
In the HBr Lewis structure, there is a single bond between the hydrogen and bromine atom, and on the bromine atom, there are three lone pairs. HBr Lewis Structure - How to Draw the Dot Structure for HBr Watch on Contents Steps #1 Draw a rough skeleton structure #2 Mention lone pairs on the atoms #3 If needed, mention formal charges on the atoms
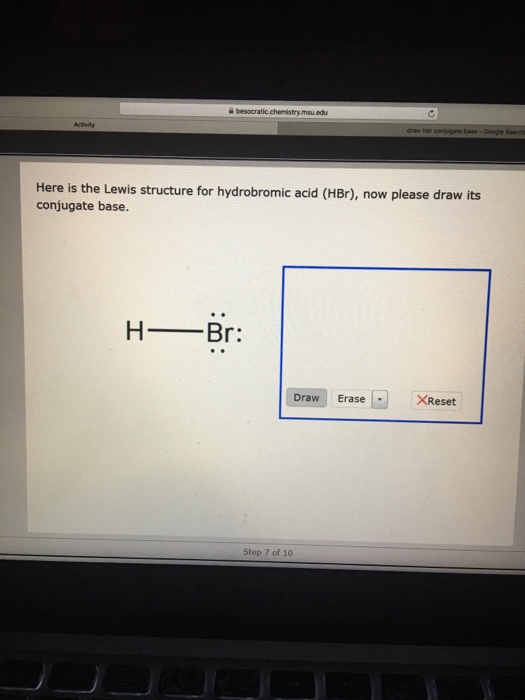
Solved Here is the Lewis structure for hydrobromic acid
Follow these simple steps to draw Lewis dot structures: Draw the atoms on paper and put dots around them to represent valence electrons of the atom. Be sure to have the correct number of electrons. If the species is an ion, add or subtract electrons corresponding to the charge of the ion. Add an electron for every negative (-) charge, and.

Hbr Lewis Structure Transborder Media
The Lewis structure of HBr contains a single bond between the hydrogen atom and bromine atom. There are three lone pairs on the bromine atom, and the hydrogen atom does not have any lone pair. HBr Lewis Structure - How to Draw the Dot Structure for HBr Watch on Contents Steps #1 Draw skeleton #2 Show chemical bond #3 Mark lone pairs

Hbr Lewis Structure Transborder Media
Connect each atom to the central atom with a single bond (one electron pair). Subtract the number of bonding electrons from the total. Distribute the remaining electrons as lone pairs on the terminal atoms (except hydrogen), completing an octet around each atom. Place all remaining electrons on the central atom.

Is HBr Polar or Nonpolar? (Hydrogen Bromide) in 2021 Molecules, Ball
Lewis structure of HBrO contains a single bond between the Hydrogen (H) & Oxygen (O) atom as well as between the Oxygen (O) and Bromine (Br) atom. The Oxygen atom (O) is at the center and it is surrounded by Hydrogen and Bromine atom. The Oxygen has 2 lone pairs and the Bromine has 3 lone pairs.

Is HBr Polar or Nonpolar? (Hydrogen bromide) YouTube
The HBr Lewis Structure represents the arrangement of atoms and bonding electrons in a molecule of hydrogen bromide (HBr). It showcases the connectivity between hydrogen (H) and bromine (Br) atoms, giving us a visual representation of their covalent bond. Atomic Information: Hydrogen (H): 1 proton, 1 electron Bromine (Br): 35 protons, 35 electrons

Lewis Structure Hbr
Step 1 To find out the Lewis Structure of any given molecule, the first step is to find out the total valence electron number. Electron loss signifies the increase of positive charge hence we use the sign '+'. The gain of electrons increases the number of negatively charged electrons therefore we use the '-' sign. Step 2
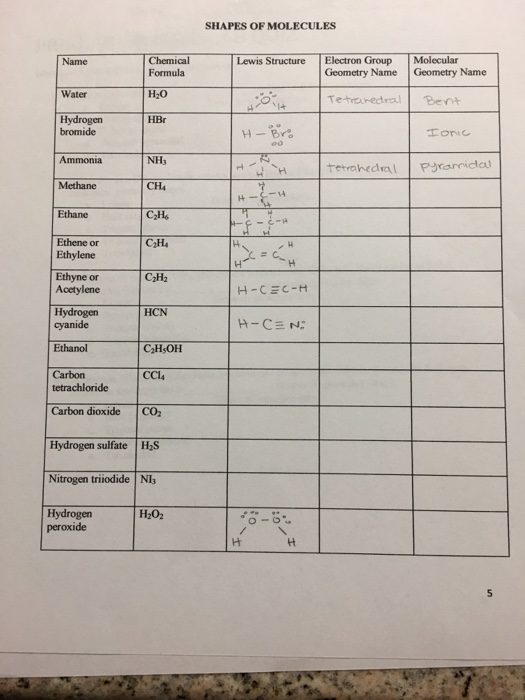
Solved SHAPES OF MOLECULES Chemical Lewis Structure Electron
Share Watch on See the Big List of Lewis Structures Transcript: Hi, this is Dr. B. Let's do the Lewis structure for HBr, hydrobromic acid. On the periodic table, Hydrogen is in group 1, so it has 1 valence electron, and Bromine is in group 7, sometimes called 17, it has 7 valence electrons; for a total of 8 valence electrons.

Hydrogen bromide (HBr) molecule. Skeletal formula Stock Vector Image
Steps to draw HBr lewis structure as follows: Determine the H and Br position in periodic table group and evaluate total valence electrons on HBr molecule. Most electronegative atom is at central position and makes bonding within H and Br atoms. After bonding remaining electrons put on bonding atoms and mark lone electron pairs.

Draw the Lewis Structure of HBr (hydrogen bromide) YouTube
Lewis structure of HBr (or Hydrogen bromide) contains one single bond between the Hydrogen (H) and Bromine (Br) atom. The Bromine atom has 3 lone pairs. Let's draw and understand this lewis dot structure step by step. (Note: Take a pen and paper with you and try to draw this lewis structure along with me.